ORGANIC CHEMISTRY : UV-VIS Spectroscopy
UV-VIS : Ultra Violet spectroscopy
Electrons in π-bonds can absorb light energy of photons to excite to higher energy levels in the molecule. Higher energy level electrons fall back to ground state releasing radiation wavelength corresponding to the energy gap, therefore p-bonds next to each other creates a good pathway or ladder by which absorbed energy from photons can excite electrons. This is called a CHROMOPHORE.
This underlying principle imparts color to some organic molecules. However, for UV- spectroscopy the molecule absorb energy from incident UV photons and emit energy in the UV region measured by a UV detector.
Common chromophores in molecules are C=C, C=O, C = C
Conjugated p-bonds creates more possibility of having allowed energy states above the ground state in which the electron can fall after excitation. This reduces the energy gap and cause emission of lower energy or longer wavelength radiation compared to the absorbed radiation energy.
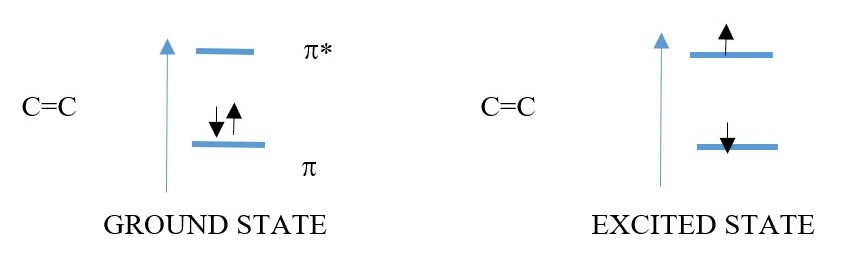
Absorbed energy is used to increase the ROTATIONAL MOTION of the vibrating molecules in UV spectroscopy. Chromophores have broad range of absorption therefore there is a maximum peak of absorption wavelength called λmax in UV spectrum of a molecule. Electrons transitions in C=C Bonds occur in π to π* energy gap HOMO to LUMO.
π = HOMO
π*=LUMO
However, carbon bonded to heteroatoms such as Oxygen, a non-bonding lone pair electron can also absorb energy and excite into a LUMO of the C=O: bond, thus a C=O have n to π* transitions. This has lower energy or longer wavelength.
π= HOMO
π*=LUMO
n = non-bonding atomic orbital of Oxygen [ contains lone pair of electrons]
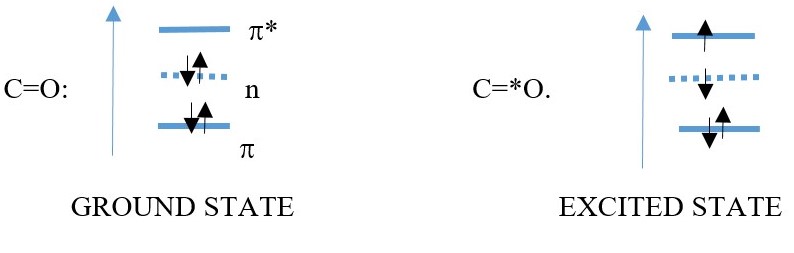
BENZENE AND DERIVATIVES
EFFECT OF SUBSTITUENTS GROUPS ON UV-VIS SPECTRUM


Electron-donating groups substituents cause red-shifting of λ max but electron-withdrawing group cause blue shift of λ max absorption.
Quantitative analysis measure concentration of compounds that absorb UV radiation because the molecules have chromophores.
Absorbance = ε c l
ε = molar absorptivity
c = molar concentration of analyte
l = path length of UV radiation through analyte