UNIT CONVERSIONS AND DIMENSIONAL ANALYSIS
Unit conversion and Dimensional analysis
in general chemistry
This deals with conversion of one unit of measurement to another unit in general chemistry. One unit of measurement can have several conversion units. For example, the mass of substance can be measured in units of grams (g). However, there are other units such as the milligrams(mg) , kilograms (kg) or the pounds( lb, lbm) . Another unit is distance or length. This can be measured in many units such as meters(m) , millimeters (mm), centimeters (cm) , inches, yards, kilometers (km) or miles.
In each conversion, it is important to know the prefix needed and how to use it to make a conversion factor or ratio. An example is given in this illustration below.
LENGTH
SI unit for length is meter.
100centimeters = 1 meter
100cm = 1m implies 1cm= (1/100) m = 0.01m = 1.0 x 10-2m
1cm=0.01m = 1.0 x 10-2m
How many meters do we have in 260cm.
1cm=1.0 x10-2m . Question is asking for meters .
Therefore, Conversion ratio (CR) = [1.0 x 10-2m/1cm] , Given amount is 260cm
Answer = Given amount x CR = 260cm x [1.0 x 10-2m/1cm] =2.6m
Other Prefixes of SI units
Decimeters 1dm = 0.1m =1x 10-1m
Milli = Millimeters 1mm=0.001m =1x 10-3m
Micro = Micrometers 1 um = 1x 10-6m ,
Nano = Nanometers 1nm = 1×10-9 m
Pico =Picometers 1pm=1 x 10-12m
Kilo = Kilometers 1km = 1 x 103m ,
Mega = megameters 1 Mm = 1 x 106m ,
Giga = gigameters 1 Gm = 1 x 109m,
Tera = terameters 1 Tm = 1 x 1012m
AREA
1cm2 = ? meters square
(1cm)2 = (1 x10-2 m)2 = 1 x10-4 m2
1cm2 = 1 x10-4 m2
VOLUME
1cm3 = ? meters cube
(1cm)3 = (1 x10-2 m)3 = 1 x10-6 m3
1cm3 = 1 x10-6 m3
MASS
SI unit for mass is kilogram.
Example convert 1.0kg into grams
1000g=1kg 1.0 x 103 g = 1.0 Kg
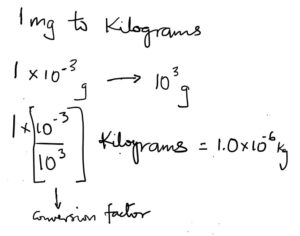
Example convert 1.0 milligrams into kilograms.
Pressure Units
Pascal (Pa)=N/m2
760mmHg=760 torrs = 1atm
How much is 540mmg in atm pressure.
ANSWER:
1atm = 760mmHg ; we are given amount in mmHg and converting into atm units.
Therefore, Conversion Ratio = [1 atm/760 mmHg] .
The given amount is 540mmHg.
Therefore Answer = CR x Given amount = 540mmHg x 1atm/760mmHg = 0.711 atm
TIME SI UNITS
60s =1minute
60min =1hour
How many seconds are there in 365 minutes.
1 minute = 60 s . question is asking for seconds , therefore CR = [60s/1minute] , Given amount is 365 Minutes
Answer = given amount x CR
=365 minutes x [60s/1 minute]
MULTIPLE UNIT CONVERSIONS
EXAMPLE:
Convert 1g/ml to kilograms/m3
ANSWER:
- You can convert 1.0 grams to kilograms.
- Kilo = 1000 , therefore the Conversion ratio = [1/1000] , amount in kilograms = (1/1000) kilograms = 0.001kg = 1.0 x 10-3kg
- Convert 1ml to cubic meters.
- 1 ml = 1 cm3 , c=centi = 1 /100 , therefore, cubic meters = (1/100)3 = (0.01) 3 = (1.0 x 10 -2)3 = 1.0 x 10-6 m3
- Divide the answer in step 1 by the answer in step 2 .
- (1 x 10-3kg) / (1 x 10-6 m3 ) = 1.0 x 103 kg/m3
EXAMPLE
Convert the pressure unit of 1.0 psi into N/m2
ANSWER:
psi refers to pounds per square inch.
Convert 1.0 pound (lb) into weight in Newtons, then convert 1.0 inches 2 into meters2 , then divide the value in Newtons by the value in meters2 .
1.0lb = 0.454 kg , therefore we convert into weight by multiplying by 9.8m/s2 . This gives 0.454 kg x 9.8m/s2 = 4.4492 kgm/s2 =4.4492 N
1inch = 0.0254 meters , therefore, (1inche)2 = (0.0254 meters )2 = 6.4516 x 10-4 m2
Dividing ; gives 4.4492 N / (6.4516 x 10-4 m2 ) = 6.9 x 103 N/m2
EXAMPLE :
Convert 23 m/s to miles/hour
ANSWER:
- Convert 23meters into miles
- Convert 1 second to hours
- Divide answer 1 by answer 2.

